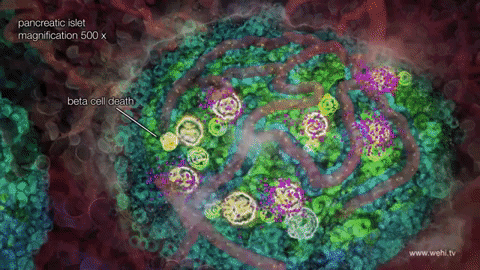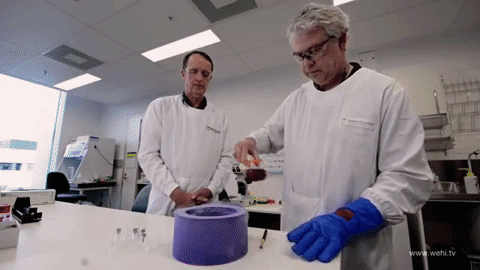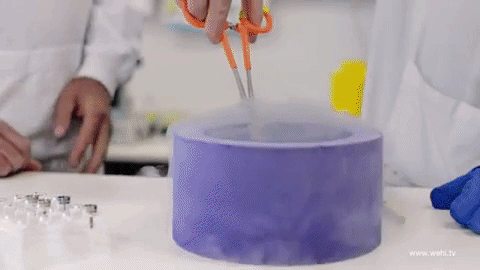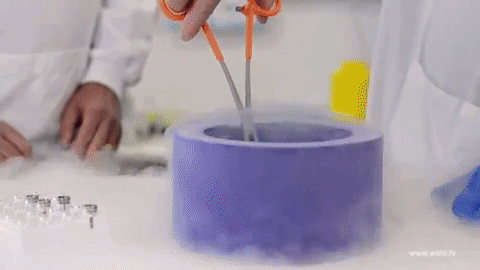
When inventor Frederick Banting discovered insulin in 1923, he refused to put his name on the patent. He felt it was unethical for a doctor to profit from a discovery that would save lives. Banting’s co-inventors, James Collip and Charles Best, sold the insulin patent to the University of Toronto for a mere $1. They wanted everyone who needed their medication to be able to afford it.
Today, Banting and his colleagues would be spinning in their graves: Their drug, which many people with diabetes rely on, has become the poster child for pharmaceutical price gouging.
The cost of the four most popular types of insulin has tripled over the past decade, and the out-of-pocket prescription costs patients now face have doubled. By 2016, the average price per month rose to $450 — and costs continue to rise, so much so that as many as one in four people with diabetes are now skimping on or skipping lifesaving doses.
The ‘big three’ insulin producers – Eli Lilly, Novo Nordisk and Sanofi – dominate more than 90% of the world insulin market by value. Often only one of these companies supplies insulin in a country, which means they more or less hold a monopoly there and can set prices as they wish. In some countries like India and China, there are domestic insulin companies that help drive down the price.
When it comes to the question of generic insulin, we are faced with another complicated issue. Insulin is a therapeutic biological product (or ‘biologic’), rather than a chemically synthesized molecule. This means it cannot be made as generic in the same way as other drugs. Creating what is called a biosimilar is a lot more complicated and expensive than just duplicating a chemical molecule.
There is a little market incentive to produce biosimilars because it costs nearly as much as making a new drug and companies must go through all the approval stages and trials that a new drug is required to go through. Not to mention, current biosimilar insulins on the market – primarily produced by the ‘big three’ – have only reduced the price by about 10-15%.
A ‘Pay for delay’ agreement is a patent dispute settlement in which a generic (in the case of insulin, a biosimilar) manufacturer acknowledges the original patent of a pharmaceutical company and agrees to refrain from marketing its product for a specific period of time. In return, the company receives a payment from the patent-holder. This means it is actually legal for one insulin producer to pay another one not to enter the market.
A few years ago the company Merck announced plans to sell a biosimilar version of Sanofi’s Lantus. Sanofi sued, and eventually, Merck announced that it was no longer pursuing its biosimilar, presumably due to payments from Sanofi to stay away. If Pay for delay schemes don’t work, the ‘big three’ can still sue other players, prolonging processes and pushing players out of the market because of legal fees and time-wasting. All of these are win-wins for companies and lose-lose for patients.
Physicians in many countries are allowed to collect fees from pharmaceutical companies for talks, advice and more. Supposedly, these are to compensate physicians for their expertise and time. However, they can create loyalty to a company and may influence prescribing habits – a belief shared by some pharmaceutical salespeople. In countries like India, physicians are allowed to sell and profit off insulin directly through patients, or through pharmacies they themselves own, cutting out middlemen and the retail pharmacies. Thus, they lose the incentive to find the lowest price insulin for their patients. Insulin companies also focus on ‘insulin-starts’, or the insulin the physician diagnosing patients begins with. As patients are reluctant to change, a number of marketing and financial incentives are employed to influence this decision.
Patients are speaking out about these issues all around the world. In the USA where prices have skyrocketed especially, T1International Chapters are being formed where patient advocates are educating and pushing for policy change.