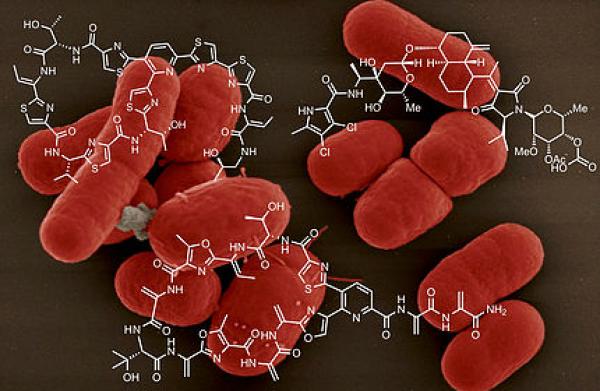
University of Colorado Boulder chemistry researchers have developed a novel way to synthesize and optimize a naturally-occurring antibiotic compound that could one day be used to fight lethal drug-resistant infections such as Staphylococcus aureus, commonly known as MRSA.
Antibiotic-resistant infections afflict over 2 million people annually and result in over 23,000 deaths in the U.S. each year, according to the Centers for Disease Control and Prevention (CDC). A 2018 study by the CDC’s European counterpart found that drug-resistant superbugs were responsible for 33,000 deaths across Europe in 2015.
Researchers have previously identified thiopeptides, a naturally-occurring antibiotic compound, as a promising avenue of study. Thiopeptides have shown some effectiveness against MRSA and certain other bacterial species in limited trials, but their structural diversity makes it difficult to synthesize the molecules at a scale large enough for therapeutic use.
To make better use of thiopeptides, CU Boulder researchers went back to basics and re-examined previous assumptions about the foundational chemical properties of these molecules.
“We re-evaluated the structural commonalities of these thiopeptides in light of current superbugs, because no one had looked at them and analyzed them in modern context,” said Maciej Walczak, lead author of the new research and an assistant professor in CU Boulder’s Department of Chemistry.
The researchers invented a new catalyst to drive reactions that facilitate the synthesis of the thiopeptides and form the essential scaffolding needed to curtail bacterial growth. Their efforts resulted in two new broadly representative antibiotics: micrococcin P1 and thiocillin I. The compounds are efficient, scaleable and produce no harmful byproducts.
“The results exceeded our expectations,” Walczak said. “It’s a very clean reaction. The only waste produced is water and the fact that this is a very green method could be important going forward as the technology scales up.”
The study, which was funded by the National Science Foundation and co-authored by CU Boulder postdoctoral researcher Siddhartha Akasapu and graduate students Aaron Hinds and Wyatt Powell, was recently published in the journal Chemical Science.
The new chemical synthesis methodology is just a starting point, Walczak said. He and his colleagues plan to use their findings as a platform for selecting and rationing parts of the thiopeptide molecules in order to optimize their properties and apply them broadly to other bacterial classes.
The antibiotic compounds will need to complete clinical trials before they can be approved for human use, a process that can take many years. Still, the need for scientific innovation in the field of antibiotic resistant superbugs is greater than ever, Walczak said.
“Multi-drug resistance is an important global health problem and it’s going to become even more so in the years to come,” he said.